RZ402
Diabetic retinopathy (DR) affects approximately one third of adults with diabetes and is the leading cause of vision loss in the working age population. Diabetic Macular Edema (DME) is a severe vision-threatening complication of DR characterized by swelling of the retina and thickening of the macula, the part of the eye that is responsible for high-resolution vision. Anti-vascular growth factor (anti-VEGF) injections into the eye are the current standard of care for DME, requiring continued administration over long periods of time to preserve vision. Due to their invasive route of administration and occasional serious side effects, there is a tendency to delay treatment until later in the disease course, and long-term compliance with eye injection regimens can be difficult for patients. Coupled with inadequate responsiveness in some patients, this leads to overall undertreatment and suboptimal vision outcomes in DME patients.
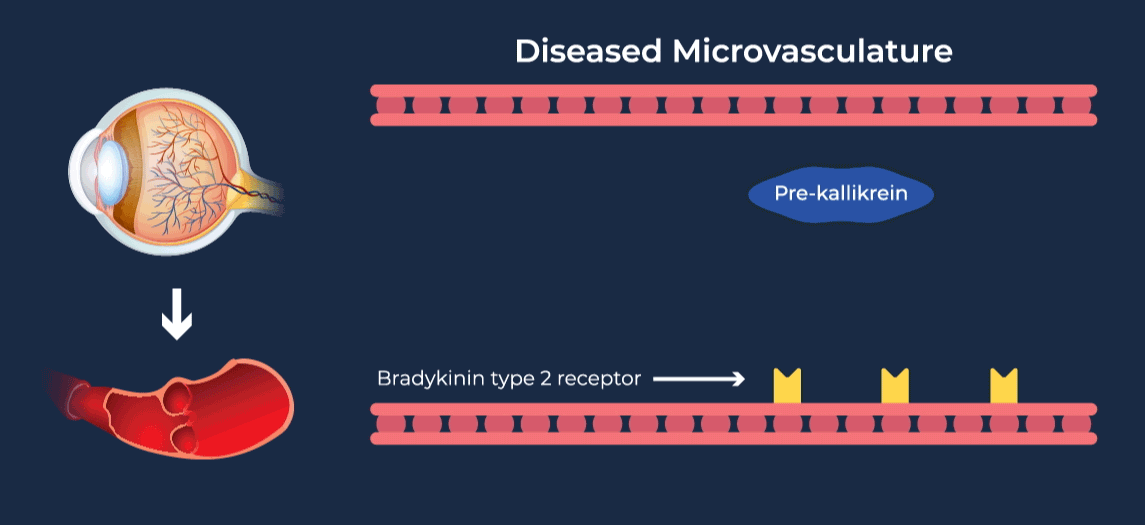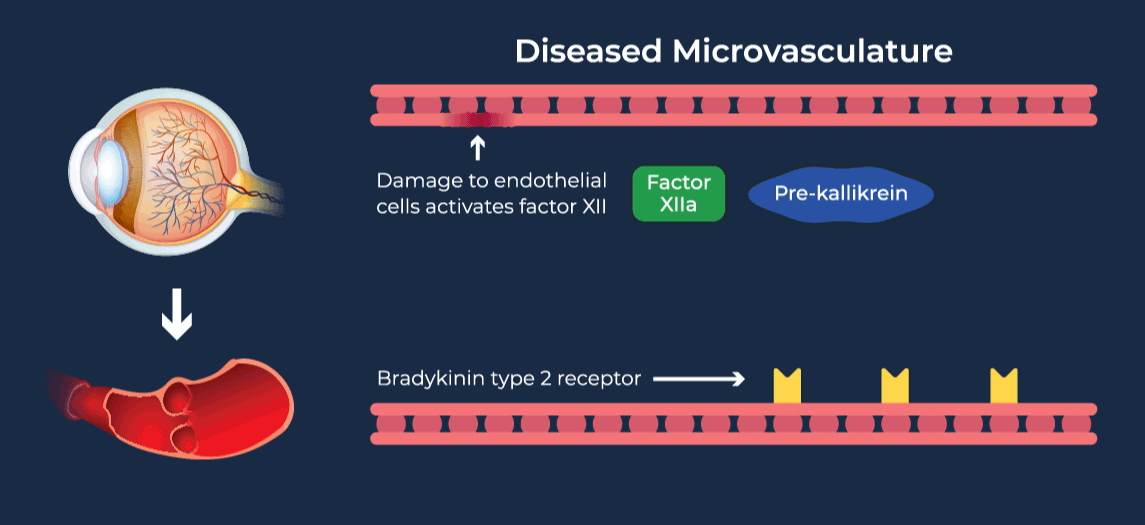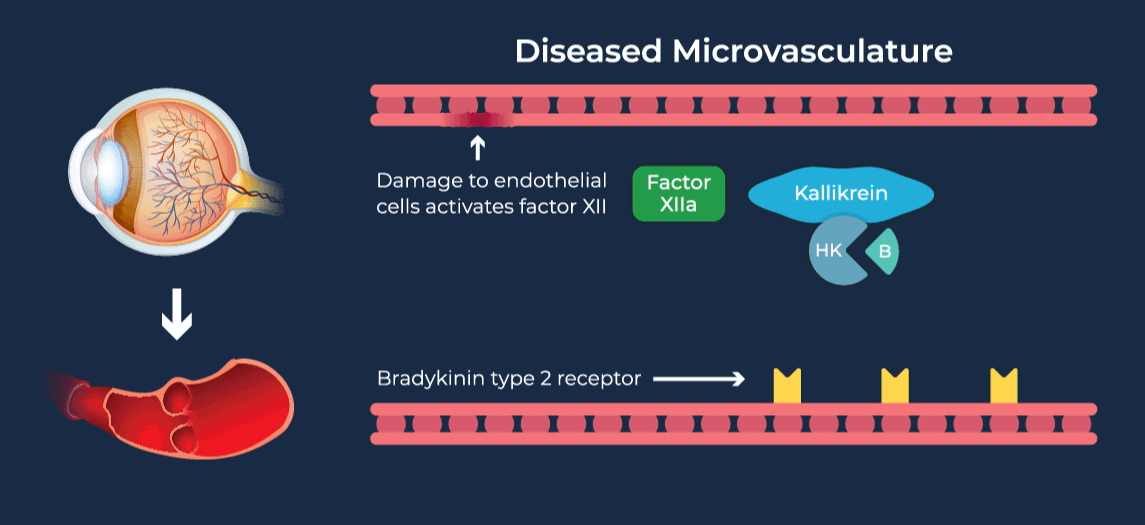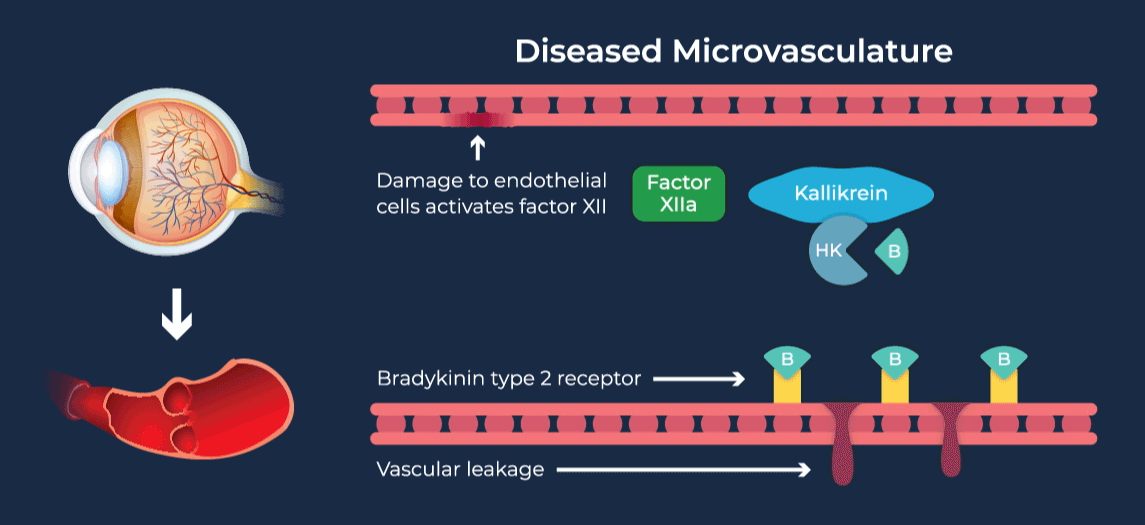
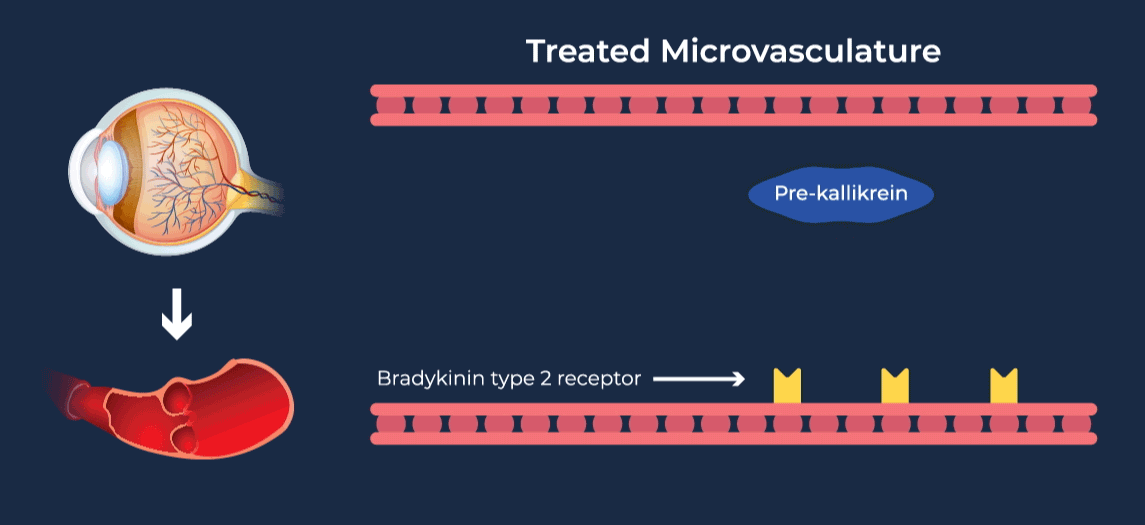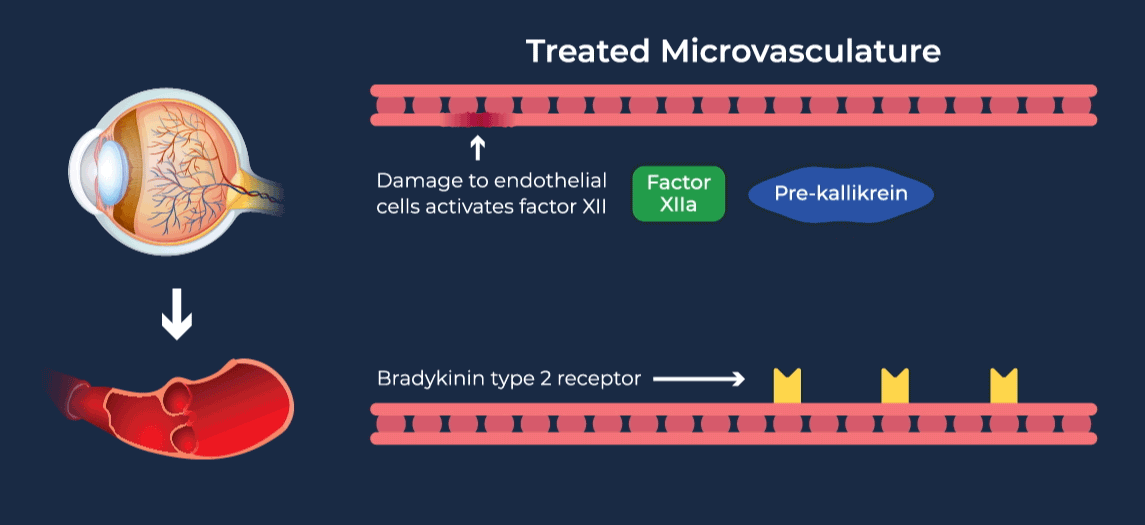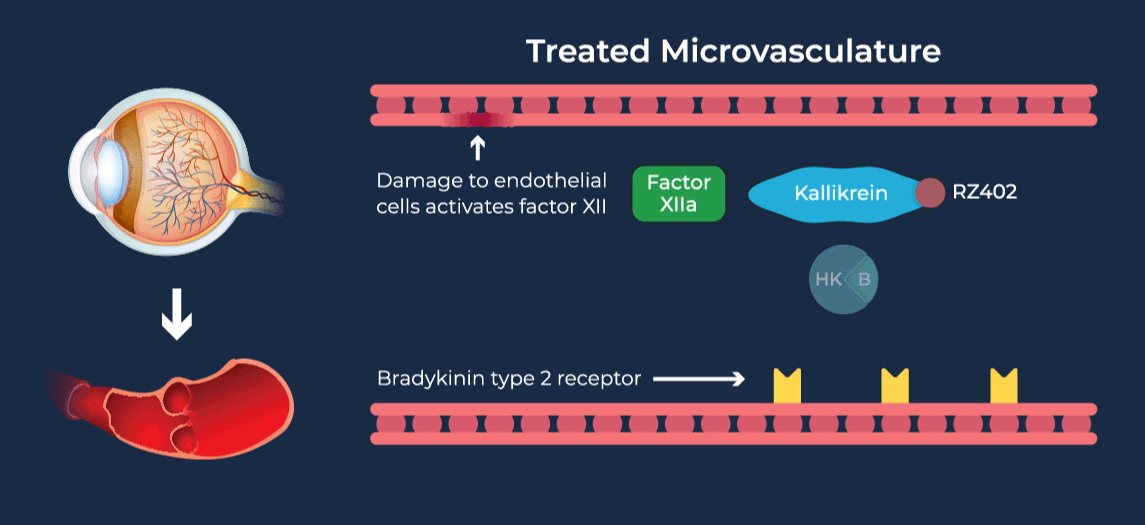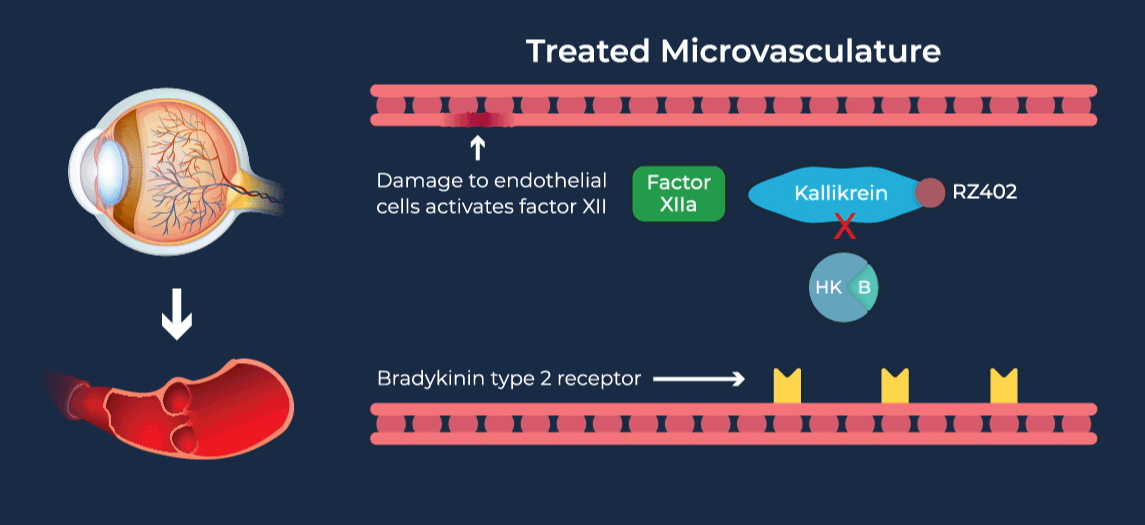
The contact-activation kallikrein-kinin system promotes increased vascular permeability and inflammation via key downstream mediators, including bradykinin, and activation of the intrinsic pathway of coagulation. Pathophysiologic upregulation of this system has been linked to a variety of diseases which are characterized by vascular dysfunction, including diabetic macular edema.
RZ402 is a small molecule selective and potent plasma kallikrein inhibitor (PKI) being developed as a potential oral therapy for the chronic treatment of diabetic macular edema (DME). By inhibiting the formation of kallikrein, RZ402 is designed to block downstream bradykinin production and the pro-inflammatory, pro-coagulant, and fluid-leakage contact-activation cascade.
RZ402 has been shown to reduce and prevent retinal vascular leakage in animal models by 80-90%.
Publications
Plasma Kallikrein Mediates Angiotensin II Type 1 Receptor–Stimulated Retinal Vascular Permeability
Read MoreNonclinical safety and pharmacology of RZ402, a plasma kallikrein inhibitor, for the treatment of diabetic macular edema as a daily oral therapy
Read MoreClinical Trials
RZ402, our oral PKI in development for DME, recently announced results from the Phase 1b study. View Results
RZ402, our oral PKI in development for DME, recently announced results from the Phase 1b study. View Results
Rezolute initiated a Phase 2a study in December 2022, with announcement of top line data planned for early 2024.
x close

