RZ358
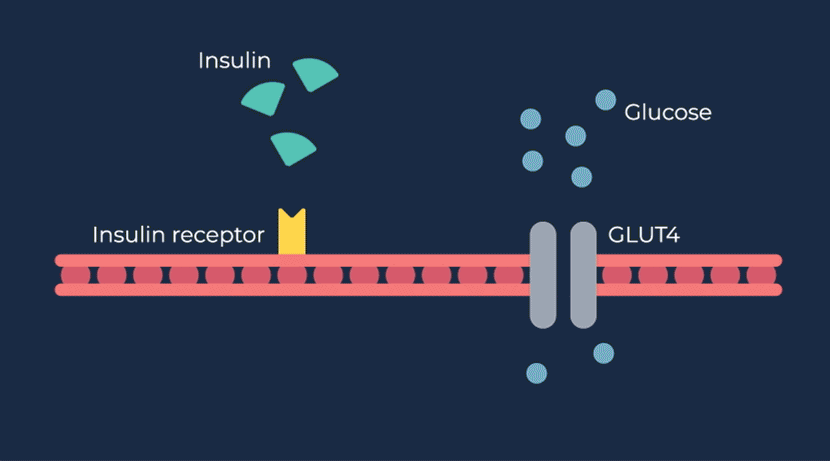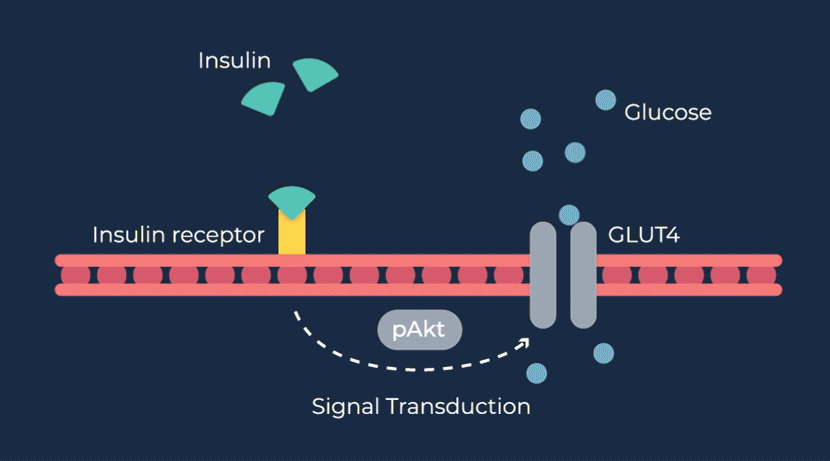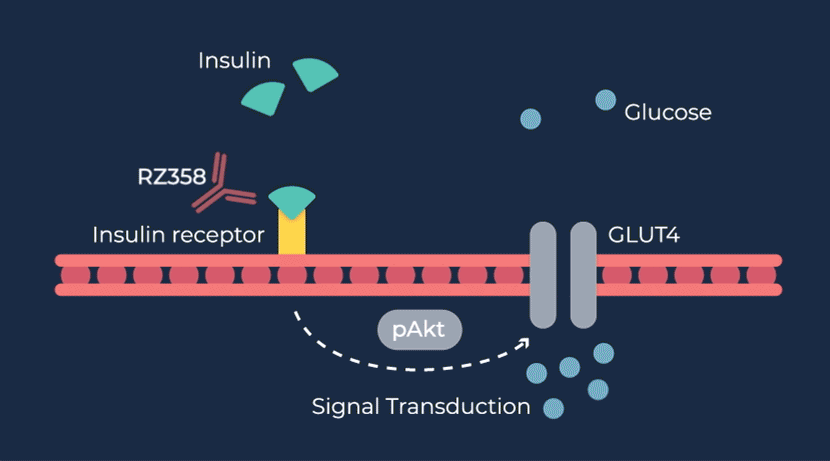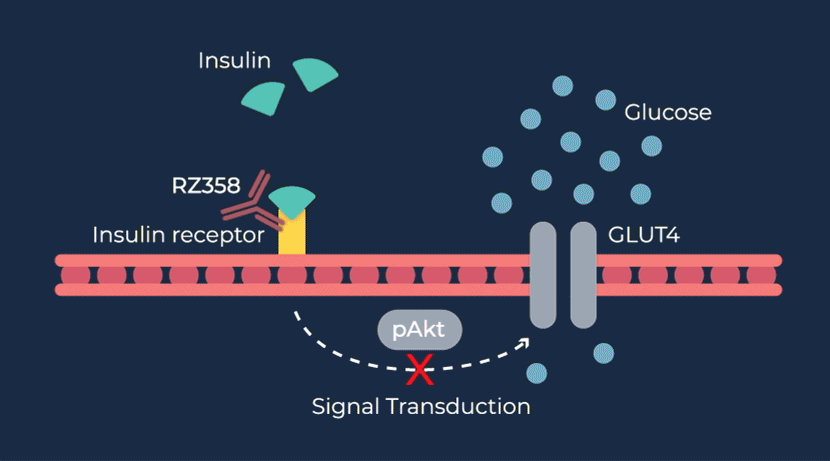
Congenital hyperinsulinism (HI) is a rare pediatric genetic disorder characterized by excessive production of insulin by the pancreas. If left untreated, elevated insulin levels can cause extreme hypoglycemic (low blood sugar) events, increasing the risk of neurologic and developmental complications, including persistent feeding problems, learning disabilities, recurrent seizures, brain damage or even death.
RZ358 is a fully human monoclonal antibody that works downstream from the pancreas and other sources of insulin or related paraneoplastic substances, and instead binds to a unique allosteric site on insulin receptors in the liver, fat, and muscle. The antibody counteracts excess insulin receptor activation by insulin and other effector substances (such as IGF-2), thereby improving hypoglycemia. Because RZ358 acts downstream from the pancreas at the insulin receptor, it has the potential to be universally effective at treating hypoglycemia due to congenital HI, regardless of the causative genetic defect, as well as acquired forms of HI such as those mediated by insulinomas (ICTs) and other tumor types (NICTs).
Publications
The birth prevalence of congenital hyperinsulinism: a narrative review of the epidemiology of a rare disease
Read MoreAn Analysis of Overnight Hypoglycemia in Patients with Congenital Hyperinsulinism:Results from the RZ358-606 (RIZE) StudyWhile
Read MoreClinical Studies
Rezolute recently revealed results from the Phase 2b study. View Results
x close


